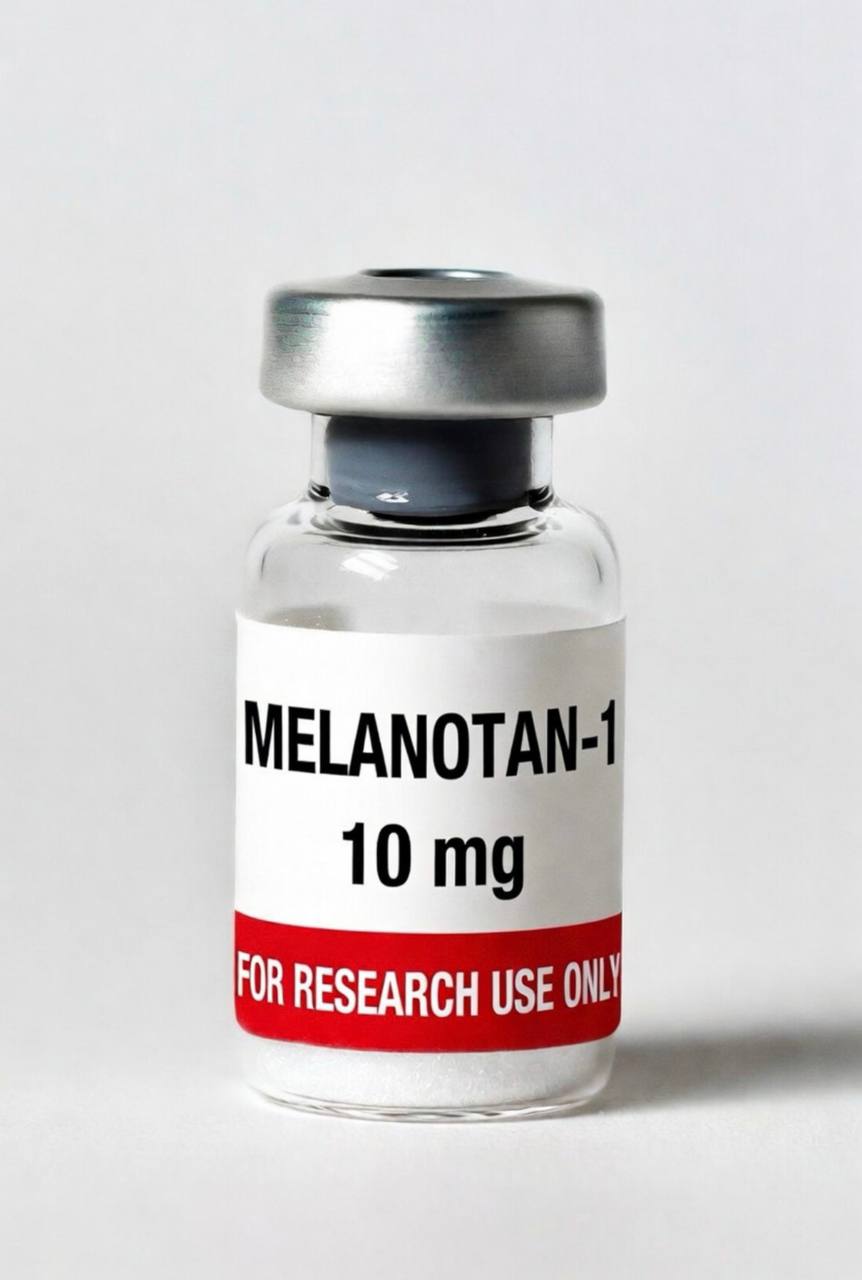
Quick Facts
- Purity: 98%+
- Form: Lyophilized powder, 10mg vial
- Storage: −20°C long-term; 2–8°C short-term. Protect from light. Avoid repeated freeze-thaw cycles.
- CAS Number: 75921-69-6
- Molecular Formula: C₇₈H₁₁₁N₂₁O₁₉
- Molecular Weight: 1646.9 g/mol
- Also Known As: Afamelanotide, [Nle4, D-Phe7]-α-MSH
- COA: Available upon request
About Melanotan-1 (Afamelanotide)
Melanotan-1, also known as Afamelanotide or [Nle4, D-Phe7]-α-MSH, is a synthetic 13-amino acid analogue of the endogenous peptide hormone alpha-melanocyte-stimulating hormone (α-MSH). The two structural modifications — substitution of methionine at position 4 with norleucine (Nle4) and substitution of L-phenylalanine at position 7 with its D-isomer (D-Phe7) — confer significantly enhanced receptor binding affinity and resistance to enzymatic degradation compared to the native α-MSH sequence.
α-MSH is derived from the precursor pro-opiomelanocortin (POMC) and exerts its biological effects primarily through the melanocortin receptor family (MC1R through MC5R). Melanotan-1 has been studied extensively as a high-affinity ligand at MC1R, which is the primary receptor mediating eumelanin production in melanocyte cell biology. MC1R activation by melanocortin ligands stimulates adenylyl cyclase via Gs coupling, elevating intracellular cAMP, which activates PKA and ultimately upregulates microphthalmia-associated transcription factor (MITF) and tyrosinase expression — the rate-limiting enzyme in melanin biosynthesis.
In research settings, Melanotan-1 has been used as a pharmacological tool to study melanocortin receptor pharmacology, the cAMP/PKA/MITF signaling cascade in melanocytes, UV-independent pigmentation mechanisms, and the role of MC1R in photoprotective biology. Its high binding affinity and extended half-life relative to native α-MSH make it a preferred research ligand for in vitro receptor studies.
Research Context
- MC1R binding pharmacology and melanocortin receptor family selectivity studies
- cAMP/PKA/MITF signaling cascade in melanocyte cell culture models
- Eumelanin vs. pheomelanin synthesis pathway regulation research
- UV-independent pigmentation and photoprotection mechanism studies
- POMC-derived peptide biology and pro-opiomelanocortin processing research
- Structure-activity relationship studies comparing MT-1, MT-2, and native α-MSH
Compliance & Safety
- Intended Use: Strictly for in vitro research, laboratory analysis, and scientific study.
- Not For: Human consumption, veterinary use, cosmetic applications, or therapeutic use.
- Handling: Protect from light during handling. Use appropriate PPE in laboratory settings.
- Buyer Responsibility: By purchasing you confirm you are a qualified researcher or institution and will comply with all applicable local, state, and federal laws.
Shipping
Orders processed within 2–5 business days. Discreet, unmarked packaging on every order. International shipping available where legally permitted.
Reviews
There are no reviews yet.